 Understanding TWIST1 and EndMT
Understanding TWIST1 and EndMT
Atherosclerosis, marked by lipid accumulation and chronic inflammation, leads to plaque formation within arterial walls. Plaque rupture can trigger heart attacks or strokes, making stability a central focus in cardiovascular research. EndMT involves endothelial cells adopting mesenchymal characteristics, traditionally linked to unstable plaques. This new study challenges that assumption.
Experimental Findings
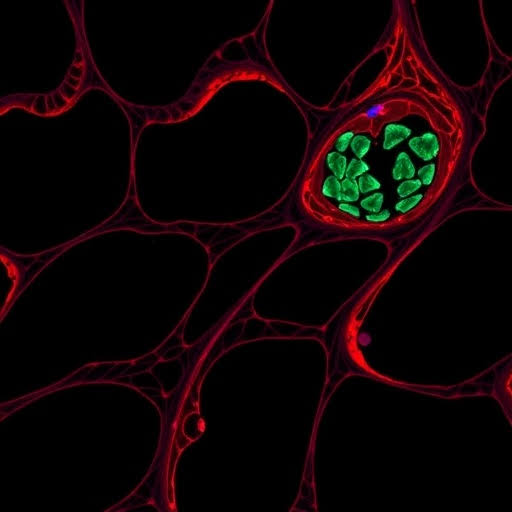
Researchers used endothelial-specific deletion of Twist1 in hypercholesterolaemic mice lacking apolipoprotein E, coupled with single-cell RNA sequencing and endothelial cell tracking. The results demonstrated that TWIST1 not only drives EndMT but also promotes features of stability, such as increased collagen deposition and higher numbers of ACTA2-positive smooth muscle-like cells. Simultaneously, necrotic core formation and macrophage infiltration were reduced.
Complementary in vitro studies on human aortic endothelial cells confirmed TWIST1’s role in cell proliferation and migration via the transcriptional coactivator PELP1. An AEBP1-dependent pathway was identified that upregulated COL4A1, further supporting endothelial proliferation and plaque reinforcement.
Implications for Cardiovascular Therapy
The findings suggest TWIST1 could be a novel therapeutic target to enhance plaque stability rather than merely reducing plaque burden. This approach may complement existing lipid-lowering and anti-inflammatory therapies, shifting the focus toward preventing rupture events. While the current evidence is preclinical, translation into human studies could open new avenues for cardiovascular disease management.
Experts caution that murine models and cell culture studies may not fully replicate human physiology. Nonetheless, combining single-cell transcriptomics with histology and functional assays provides a strong mechanistic understanding of TWIST1’s role in atherosclerosis.